
elegans, mouse, and human data suggest that hub connectivity is associated with a distinct transcriptional signature related to metabolic function 7, 11, 23, 24, 25. Preliminary evidence from human twin research suggests that certain properties of hub functional connectivity are strongly heritable 22, and analyses of C. However, it is unknown whether genetic influences are preferentially exerted across specific classes of connections, such as the costly and functionally valuable links between network hubs. 
In the developing brain, neurons can innervate precise targets, even over long anatomical distances, by following genetically regulated molecular cues 20, 21. What influences the way in which hub regions connect to each other? The rapid evolutionary expansion of network hubs in humans, coupled with evidence supporting the heritability of many different aspects of brain organization 19, suggests an important role for genes. These findings support the view that rapid expansion of multimodal association hubs, and the costly, valuable rich-club connections between them, underlies the enhanced cognitive capacity of humans compared to other species 18. They also show greater topological centrality and evolutionary divergence in the human connectome when compared to chimpanzee 17. These brain regions are disproportionately expanded in individuals with larger brains 15 and in human compared to nonhuman primates 16. Paralimbic and association hubs of the human brain also show marked inter-individual variability in connectivity and function that relates to a diverse array of behaviors 6, 12, 13, 14. In the human cortex, hubs are predominantly located in transmodal paralimbic and association areas 6, 9 and are among the most metabolically expensive elements of the connectome 10, with rich-club connections between hubs accounting for a disproportionate fraction of axonal wiring costs 3, 4, 5, 7, 11. These hubs are more strongly interconnected with each other than expected by chance, forming a rich-club 3, 4, 5, 7 that is topologically positioned to integrate functionally diverse neural systems and to mediate a large proportion of inter-regional communication 5, 8. Numerous studies, conducted in species as diverse as the nematode Caenorhabditis elegans, mouse, macaque, and human, and at scales ranging from the cellular to the macroscopic, have shown that this complex organization is, in part, attributable to a heterogeneous distribution of connectivity across neural elements, such that a large fraction of network connections is concentrated on a small subset of network nodes called hubs 3, 4, 5, 6, 7. Nervous systems are intricately connected networks with complex wiring patterns that are neither completely random nor completely ordered 1, 2. Our findings indicate that genes play a strong and preferential role in shaping the functionally valuable, metabolically costly connections between connectome hubs. Finally, comparing over thirteen generative models of network growth, we show that purely stochastic processes cannot explain the precise wiring patterns of hubs, and that model performance can be improved by incorporating genetic constraints. 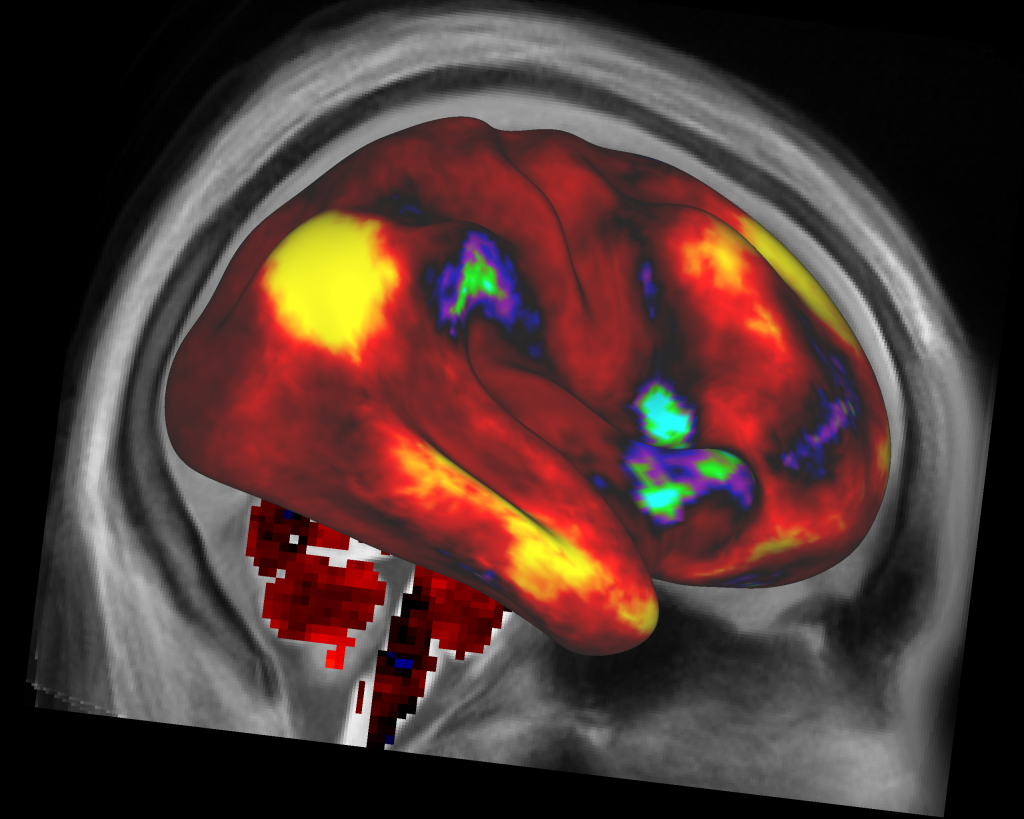
Using transcriptomic atlas data, we show that connected hubs demonstrate tight coupling of transcriptional activity related to metabolic and cytoarchitectonic similarity. Using diffusion-weighted magnetic resonance imaging in twins, we identify a major role for genes, showing that they preferentially influence connectivity strength between network hubs of the human connectome. The mechanisms driving this organization are poorly understood. 
Brain network hubs are both highly connected and highly inter-connected, forming a critical communication backbone for coherent neural dynamics.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
